Glo1 protein: glyoxalase 1
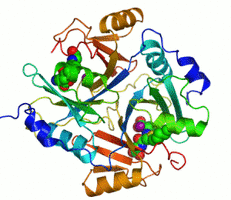
Figure 1. 3D structure of human glyoxalase 1
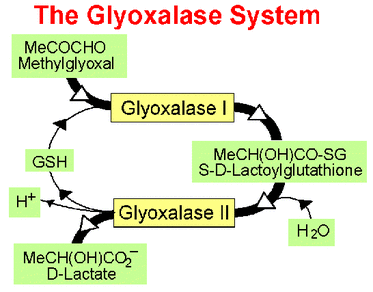
Glyoxalase I (also known as lactoylglutathione lyase) is part of the glyoxalase system (Fig. 2), a two-step system for detoxifying methylglyoxal, a side product of glycolysis. This system is responsible for the conversion of reactive, acyclic alpha-oxoaldehydes into the corresponding alpha-hydroxyacids. Eukaryotic glyoxalase I prefers the divalent cation zinc as cofactor. Human glyoxalase I is a two-domain enzyme and it has the structure of a domain-swapped dimer with two active sites located at the dimer interface. In yeast, in various plants, insects and Plasmodia, glyoxalase I is four-domain, possibly the result of a further gene duplication and an additional gene fusing event. [1]
glyoxalase 1 in Humans (Homo sapiens)
Ontology - UniProt Information [3]
Subunit structure: Homodimer
Cofactor: One zinc ion per subunit
Post-translational modification:
1) Glutathionylation at Cys-139 inhibits enzyme activity
2) Exists in a nitric oxide (NO)-modified form. The exact nature of the modification is unknown, but it suppresses the TNF-induced transcriptional activity of NF-kappa-B
Polymorphism: Exists in three separable isoforms which originate from two alleles in the genome. These correspond to two homodimers and one heterodimer composed of two subunits showing different electrophoretic properties. Isoform 2 is missing amino acids 105-119.
Biological process: carbohydrate, glutathione, and methylglyoxal metabolic process, negative regulation of apoptotic process, regulation of transcription from RNA polymerase II promoter
Cellular component: cytoplasm
Molecular function: lactoylglutathione lyase activity, metal ion binding
Cofactor: One zinc ion per subunit
Post-translational modification:
1) Glutathionylation at Cys-139 inhibits enzyme activity
2) Exists in a nitric oxide (NO)-modified form. The exact nature of the modification is unknown, but it suppresses the TNF-induced transcriptional activity of NF-kappa-B
Polymorphism: Exists in three separable isoforms which originate from two alleles in the genome. These correspond to two homodimers and one heterodimer composed of two subunits showing different electrophoretic properties. Isoform 2 is missing amino acids 105-119.
Biological process: carbohydrate, glutathione, and methylglyoxal metabolic process, negative regulation of apoptotic process, regulation of transcription from RNA polymerase II promoter
Cellular component: cytoplasm
Molecular function: lactoylglutathione lyase activity, metal ion binding
References
[1] NCBI. U.S. National Library of Medicine, n.d. Retrieved February 13, 2013 from http://www.ncbi.nlm.nih.gov/Structure/cdd/wrpsb.cgi?INPUT_&SEQUENCE=NP_006699.2
[2] "Glyoxalase System." Clinical Sciences Research Institute. Warwick. Retrieved February 28, 2013 from http://www2.warwick.ac.uk/fac/med/research/csri/proteindamage/researchinterest/glyoxalase_i_inhibitors/
[3] http://www.uniprot.org/uniprot/Q04760
[2] "Glyoxalase System." Clinical Sciences Research Institute. Warwick. Retrieved February 28, 2013 from http://www2.warwick.ac.uk/fac/med/research/csri/proteindamage/researchinterest/glyoxalase_i_inhibitors/
[3] http://www.uniprot.org/uniprot/Q04760
Site created by: Emma Baar
Last updated: 5-14-2013
University of Wisconsin - Madison
Last updated: 5-14-2013
University of Wisconsin - Madison